

17075 Devonshire St., Suite 205 Northridge, CA 91325
16260 Ventura Blvd., Suite 600 Encino, CA 91436
555 Marin St., Suite 110 Thousand Oaks, CA 91360
Mon - Fri : 9:00 AM - 5:00 PM

The only practice in the San Fernando Valley offering BLVR with the FDA-approved Zephyr Endobronchial Valve System
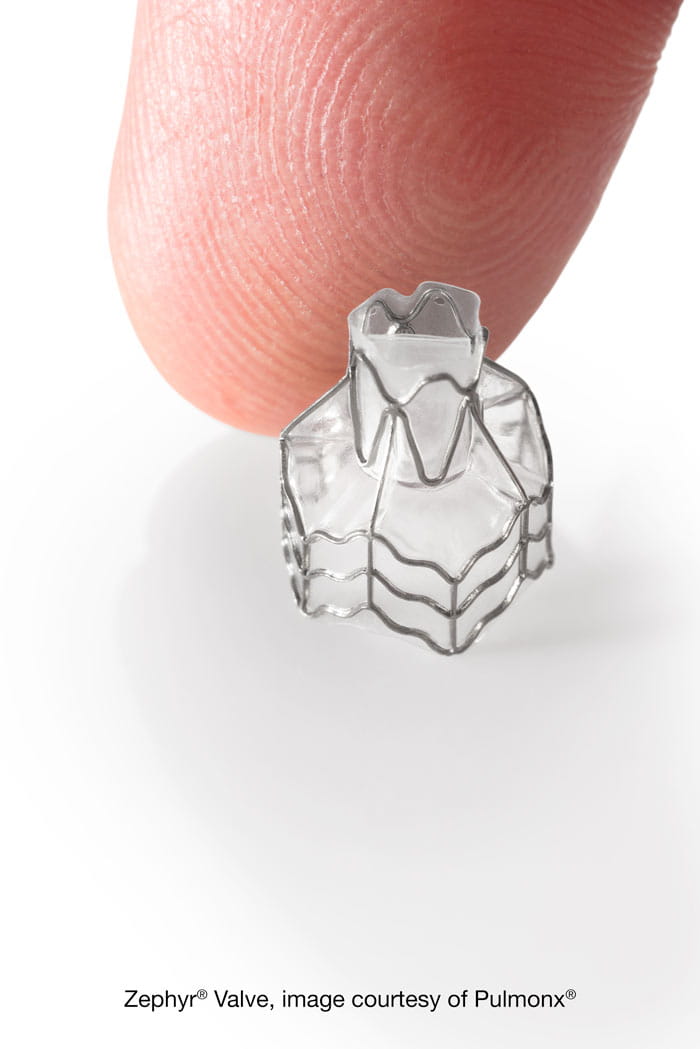
Bronchoscopic Lung Volume Reduction, or BLVR, is an FDA-approved, minimally invasive bronchoscopic procedure for patients with severe emphysema who continue to struggle with breathlessness despite optimal medical therapy. PulmoCrit Associates is the only practice in the San Fernando Valley offering this technology using the Zephyr Endobronchial Valve System by Pulmonx — bringing a treatment previously available only at major academic medical centers directly to our community.
For patients who qualify, BLVR can produce meaningful, lasting improvements in lung function, exercise capacity, and quality of life — without surgery, without incisions, and with the ability to reverse the procedure if needed.
Zephyr® Valve image courtesy of Pulmonx®
Emphysema is a progressive form of COPD in which the air sacs of the lungs (alveoli) are permanently damaged, losing their normal elasticity. As the disease advances, damaged air sacs merge into larger, inefficient spaces that trap air — a condition called hyperinflation. The lungs become overinflated, the diaphragm is pushed downward and flattened, and every breath requires more effort than a healthy lung would demand.
This trapped air creates a cascade of problems: the healthier portions of the lung are compressed and cannot expand fully, breathing muscles become chronically fatigued, and the patient feels chronically short of breath even at rest or with minimal activity. Conventional bronchodilator medications can help, but they do not address the root mechanical problem of the trapped, over-expanded diseased lobe.
BLVR targets this mechanical problem directly — by selectively collapsing the most diseased, hyperinflated lobe, BLVR restores space for healthier lung tissue to expand and allows the diaphragm to return to a more functional position.
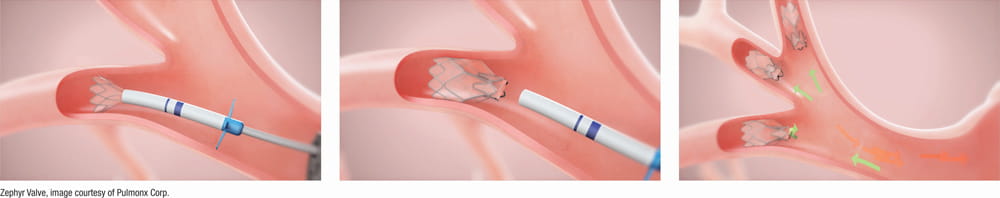
Zephyr® Valve procedure illustration courtesy of Pulmonx Corp.
The Zephyr Valve is a small, one-way valve made of a nitinol (shape-memory metal) frame covered with a biocompatible polymer membrane. Each valve is approximately the diameter of a pencil eraser. During the procedure, 3 to 5 valves are placed bronchoscopically — through the mouth, down the airways, and into the segmental bronchi (airway branches) leading to the most diseased lobe of the lung.
Once in place, the valves function as a one-way gate: they allow air and secretions to exit the diseased lobe on exhalation, but block air from entering on inhalation. Over the days and weeks following the procedure, the trapped air gradually escapes, and the diseased lobe slowly deflates and collapses. This process — called lobar volume reduction — frees up space within the chest for the remaining, healthier lung tissue to expand and function more efficiently. The result is an improvement in lung mechanics, reduced hyperinflation, and, for many patients, meaningful relief from breathlessness.
The valves are permanent implants but are entirely reversible — they can be removed bronchoscopically at any time if needed.
BLVR is not appropriate for all patients with COPD. Careful evaluation and patient selection are essential to achieving good outcomes. Candidates typically meet all of the following criteria:
A thorough evaluation at PulmoCrit includes CT scan review, complete pulmonary function tests, a 6-minute walk test, echocardiogram, and a detailed clinical assessment to determine whether you are an appropriate candidate for this procedure.
Collateral ventilation (CV) refers to airflow that passes between lung segments through pathways other than the normal airways. In patients with significant collateral ventilation in their target lobe, air enters the lobe through these alternative pathways even after valves are placed — preventing the lobe from deflating and making the procedure ineffective.
CV status is assessed during the bronchoscopic procedure using the Chartis Pulmonary Assessment System, a specialized catheter that is placed into the targeted lobe bronchus and measures whether air flow continues after the airway is blocked. Patients who are CV-negative — meaning little or no air flows through collateral pathways — are ideal candidates and the ones most likely to achieve meaningful lobar volume reduction. Patients who are CV-positive are generally not candidates for Zephyr valve therapy.
This step is a critical part of the procedure and one reason that BLVR requires specialist expertise in advanced bronchoscopy — the assessment must be performed accurately in order to select the right patients and achieve the best outcomes.
The Zephyr Endobronchial Valve received FDA approval in June 2018 based largely on the results of the LIBERATE trial — a multicenter, prospective, randomized controlled trial that evaluated BLVR in severe emphysema patients. Key findings at 12 months included:
These results represent a clinically meaningful benefit for a patient population that has historically had few effective treatment options beyond maximizing inhaler therapy and pulmonary rehabilitation.
Before the Procedure: You will undergo a comprehensive evaluation including review of CT imaging, pulmonary function tests, a 6-minute walk test, and other assessments. If you are a candidate, the procedure is scheduled and performed at a hospital where our team holds active privileges. You will be instructed to fast for at least 8 hours beforehand and will need to arrange for someone to drive you home. Blood-thinning medications may need to be temporarily held — your physician will provide specific instructions.
During the Procedure: BLVR is performed under general anesthesia or deep sedation. A flexible bronchoscope is passed through the mouth into the airways. The Chartis catheter is first used to assess collateral ventilation in the target lobe. If the assessment confirms CV-negative status, the delivery catheter is used to place 3 to 5 Zephyr valves into the segmental airways of the target lobe. Each valve expands and anchors into the airway wall. The entire procedure typically takes 30 to 60 minutes.
After the Procedure — Monitoring Period: Because BLVR can trigger a pneumothorax (collapsed lung) as the treated lobe begins to deflate, patients are hospitalized and monitored closely for 48 to 72 hours following valve placement. This monitoring period is a routine and planned part of the procedure. If a pneumothorax occurs, it is managed with a chest tube and typically resolves without further intervention. Most patients are discharged within 2 to 4 days.
Recovery and Results: The treated lobe does not collapse immediately — volume reduction occurs gradually over the first weeks to months following the procedure. Follow-up CT imaging is typically performed at 1 to 3 months to confirm lobar collapse. Patients generally begin to notice improvement in breathlessness and exercise tolerance over this period. Most improvement is established by 3 to 6 months and is durable at 12 months and beyond in appropriately selected patients.
BLVR with the Zephyr valve is a well-studied procedure with a defined and manageable risk profile. Patients should be aware of the following:
Our team's experience in advanced bronchoscopy and hospital-based critical care means that complications — if they occur — are managed promptly and effectively.
BLVR is a specialized procedure that requires a unique combination of advanced bronchoscopic skill, experience with patient selection, and the ability to manage post-procedural complications. Most community pulmonary practices are not equipped to offer it — and within the San Fernando Valley, PulmoCrit Associates is the only group that does.
Dr. Kasra Sedarati leads our BLVR program. His high-volume bronchoscopy practice — which includes robotic bronchoscopy, EBUS, navigational bronchoscopy, and cryobiopsy — provides the procedural foundation that BLVR demands. Our physicians also hold active attending roles at regional hospitals, ensuring patients receive appropriate monitoring and management in the post-procedural period.
If you or a loved one has severe emphysema and has been told there are no further treatment options, BLVR may be worth exploring. We encourage you to schedule an evaluation so we can review your history, imaging, and pulmonary function data and determine whether you are a candidate for this procedure.
Interested in BLVR? Request an Evaluation
We'll review your records and imaging and determine whether you're a candidate · Northridge & Encino