If you have severe emphysema and still struggle to breathe despite inhalers, pulmonary rehabilitation, and supplemental oxygen — you may have wondered whether anything else can be done. For many years, the answer was surgery or nothing. But that's changed.
A minimally invasive bronchoscopic procedure called Bronchoscopic Lung Volume Reduction (BLVR) using the Zephyr® Endobronchial Valve System is now FDA-approved and has been proven to significantly improve breathing in carefully selected patients with severe emphysema. And PulmoCrit Associates is the only practice in the San Fernando Valley currently offering this technology.
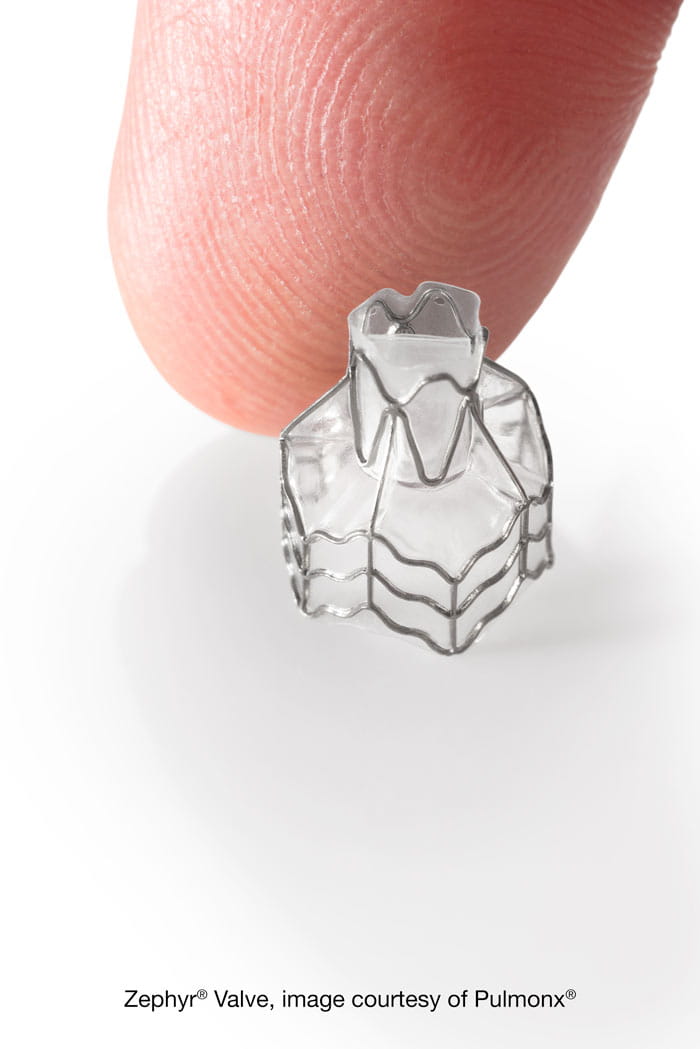
Zephyr® Valve image courtesy of Pulmonx®
What Is Emphysema — And Why Is It So Hard to Treat?
Emphysema is a progressive form of COPD in which the tiny air sacs of the lung (alveoli) are destroyed over time, most commonly from years of cigarette smoking. As these air sacs break down, they lose their elasticity and merge into large, inefficient pockets of trapped air called bullae. The lung becomes hyperinflated — overfilled with stale air that can't be pushed out — and the diaphragm flattens, making every breath feel labored.
Standard treatments — bronchodilator inhalers, inhaled steroids, pulmonary rehabilitation — help manage symptoms but cannot reverse the underlying structural damage. For patients with the most severe disease, these therapies eventually hit a ceiling. Breathing remains difficult. Exercise tolerance stays limited. Quality of life suffers.
Historically, the only surgical option offering meaningful improvement was Lung Volume Reduction Surgery (LVRS), in which a surgeon removes the most damaged portions of lung tissue. LVRS can be effective, but it carries significant surgical risk and is not appropriate for many patients. For years, there was no good middle ground.
What Is Bronchoscopic Lung Volume Reduction?
BLVR is a bronchoscopic procedure — meaning it's performed through the airways, without any incisions. The pulmonologist uses a flexible bronchoscope passed through the mouth and into the lung to place small one-way valves directly into the airways leading to the most diseased portions of the lung.
These valves — the Zephyr® Endobronchial Valves made by Pulmonx — allow air and mucus to exit the targeted lobe but prevent air from re-entering. Over time, the blocked lobe collapses and deflates. This is intentional: as the most damaged, hyperinflated tissue deflates, the healthier surrounding lung tissue has more room to expand. The diaphragm regains a more natural position. Breathing becomes easier.
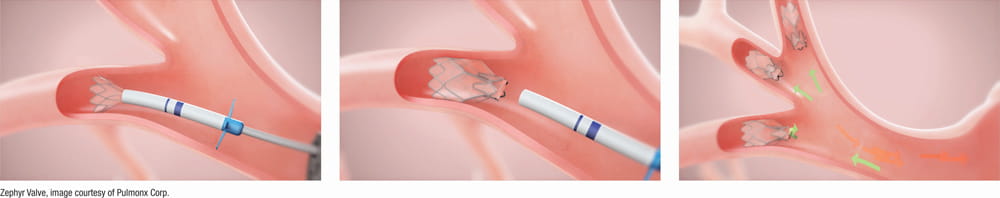
Zephyr® Valve procedure illustration courtesy of Pulmonx Corp.
How the Zephyr® Valve Works
The Zephyr Valve is a small, self-expanding device placed inside a bronchial airway using a standard bronchoscope under general anesthesia or deep sedation. It acts as a one-way gate: air and secretions can exit the diseased lobe, but air cannot flow back in. With no incoming air, the lobe progressively deflates, reducing hyperinflation and allowing the rest of the lung to work more effectively.
What Does the Clinical Evidence Show?
The strongest evidence for BLVR with the Zephyr valve comes from the landmark LIBERATE trial — a randomized, controlled study that led to FDA approval in 2018. The results were striking:
Beyond lung function, BLVR patients in the LIBERATE trial showed significant improvements in exercise capacity (6-minute walk distance), quality of life scores, and breathlessness ratings — all of which matter enormously to patients living with severe emphysema. The Zephyr valve received FDA approval in June 2018 based on this data.
Who Is a Candidate for BLVR?
BLVR is not for everyone with emphysema. It requires careful patient selection to achieve the best outcomes. Candidates typically have:
- Severe emphysema with significant hyperinflation (air trapping), confirmed on pulmonary function testing and CT scan
- Lung function in the moderate-to-severe range (FEV₁ typically 15–45% predicted) — sick enough to benefit, but not so far advanced that benefit is unlikely
- Disease that is predominantly in one lobe or region of the lung, allowing targeted treatment
- Ongoing symptoms despite maximal medical therapy (bronchodilators, pulmonary rehab)
- No significant COPD exacerbation in the preceding 3 months
- No active smoking (patients should be tobacco-free)
The Key Factor: Collateral Ventilation
The single most important determinant of BLVR success is a concept called collateral ventilation (CV). In a normal lung, each lobe is essentially isolated — air in one lobe can't flow directly into an adjacent one. In emphysema, the walls between neighboring air sacs and lobes can break down, creating pathways for air to flow between lobes. This is collateral ventilation.
If a patient has high collateral ventilation in the target lobe, air will continue to flow into that lobe through these pathways even after valves are placed — and the lobe won't deflate. In these patients, BLVR with valves simply doesn't work.
Patients with low or absent collateral ventilation — meaning the target lobe is functionally isolated — are the ones who respond to BLVR. In them, the valves can fully block airflow in, the lobe deflates, and the patient experiences meaningful improvement.
Chartis Assessment: During the bronchoscopic evaluation, your pulmonologist uses the Chartis Pulmonary Assessment System to measure collateral ventilation in real time. This straightforward measurement — performed through the bronchoscope — is the single most important step in determining whether BLVR will work for you. Only patients confirmed to have low collateral ventilation proceed to valve placement.
What to Expect: The BLVR Procedure
BLVR is performed as an outpatient or short-stay bronchoscopic procedure. Here's what the process looks like:
- Comprehensive evaluation — Your PulmoCrit pulmonologist reviews your CT scan, pulmonary function tests, and overall clinical picture to determine whether you're an appropriate candidate.
- Collateral ventilation assessment — During a bronchoscopy under sedation, the Chartis system measures airflow in the target lobe to confirm low collateral ventilation. This may be done at the same session as valve placement or as a separate visit.
- Valve placement — If the CV assessment confirms candidacy, your pulmonologist places the Zephyr valves through the bronchoscope into the airways of the target lobe. No incisions are made. Typically 3–5 valves are placed depending on the anatomy.
- Post-procedure monitoring — You'll be observed for 1–3 days, as the target lobe begins to deflate. Some patients experience transient coughing or chest discomfort as the lobe collapses — this is expected.
- Follow-up — CT scan and pulmonary function tests in the weeks and months following the procedure confirm response. Improvement in symptoms typically develops over weeks as the lung reorganizes.
What Are the Risks?
BLVR is considerably safer than lung volume reduction surgery, but it is not without risks. The most important potential complication is a pneumothorax (collapsed lung) — which can occur in roughly 20–30% of cases as the target lobe deflates rapidly. Most pneumothoraces are managed with a temporary chest tube and resolve completely. Other possible complications include COPD exacerbation, respiratory infection, and (rarely) valve migration requiring repositioning. Your PulmoCrit team will review all risks in detail during your consultation and ensure you're managed in an appropriate setting if a complication occurs.
Why PulmoCrit for BLVR?
BLVR requires a unique combination of advanced bronchoscopic skill, precise patient selection, and a team that understands emphysema management at every level. At PulmoCrit, we are the only practice in the San Fernando Valley with this capability — and our pulmonologists have the procedural expertise and clinical judgment to select the right patients and perform this procedure safely.
Dr. Kasra Sedarati leads our BLVR program. Quadruple board certified in Pulmonary, Critical Care, Sleep Medicine, and Internal Medicine, Dr. Sedarati trained at Cedars-Sinai Medical Center and brings high-volume bronchoscopic expertise — including robotic bronchoscopy, EBUS, and now BLVR — to patients in the San Fernando Valley who previously had no local access to this level of care.
You don't have to travel to Los Angeles or a major academic center for this. PulmoCrit brings the highest level of advanced pulmonary care directly to the Valley — including BLVR, robotic bronchoscopy, EBUS, and a comprehensive emphysema management program.
Is BLVR Right for You?
If you have severe emphysema and feel like you've exhausted the standard options, BLVR may be worth discussing with a specialist. Not everyone qualifies — but for those who do, the improvement in breathing, exercise tolerance, and quality of life can be life-changing.
The only way to know is an evaluation. Our team will review your existing pulmonary function tests and CT imaging and give you a straightforward answer about whether you're a candidate. If you are, we'll walk you through every step of what to expect.
Ask About BLVR at PulmoCrit
We're the only practice in the San Fernando Valley offering Bronchoscopic Lung Volume Reduction with the FDA-approved Zephyr® Endobronchial Valve. If you have severe emphysema and want to explore your options, schedule a consultation with our team today.
Request a Consultation Call (844) 428-5864